The sun provides a huge potential source of energy for our planet. Average solar energy falling on the planet is around 1300 W/m2. This is roughly the equivalent energy to that used by an electric kettle. Whilst the light the sun provides is really useful for seeing and helping plants to grow, it is not that useful for powering our modern society.
Solar Panels can help here – a solar panel converts light into electricity, which can then be used to power many things. This guide assumes a basic knowledge of electricity.
You can download a PDF of this guide here.
Solar History:
Solar Cells have been around for over 130 years! Solar cells generate electricity by absorbing the energy from light and using this to push electrons along the wires. This is called the photovoltaic effect, which was first demonstrated in 1839 by Edmond Becquerel.
Charles Frits built the first practical solar cell in 1884, using a thin covering of gold leaf over selenium. It was not very efficient, converting around 1% of the light energy to electricity, but it was a revelation at the time, with Frits stating that it produced a current “that is continuous, constant, and of considerable force”.

The development of the solar cell did not really take off until the beginning of the semiconductor industry. Transistors, and modern solar cells, both rely on the P-N Junction that can be created in a semiconductor.
What is a Semiconductor?
Just as metals conduct electricity, and are called conductors, and insulators, such as plastics, do not, there are a few elements which conduct electricity only partially, or only under certain circumstances.
These elements are called semiconductors. Silicon, Germanium and Selenium are semiconductors. The properties of a semiconductor, such as how it conducts, can be altered by adding small amounts of other elements. This is called ‘Doping’, and changes the semiconductor to P-Type (where there are more spaces electrons can flow into) or N-Type (where there are more electrons than normal).
A lot of modern semiconductor devices, such as diodes, transistors and solar panels, rely on a property of semiconductors which occurs when a P-type and an N-type semiconductor are joined. The PN Junction which is formed can be thought of as like a cliff – electrons can travel from the high side over the cliff to the low side, but cannot get from the low side back up. This property is why a diode will only conduct in 1 direction.
Modern Solar Cells:
The modern solar cell was patented in 1946 by Russell Ohl (US Patent 2,402,662) following work on semiconductor impurities and his discovery of the PN junction (in 1939). Solar cells can be made from most semiconductors, but the most common currently used is silicon. This is because it has a good price / performance trade off. Gallium Arsenide is used for higher performance solar cells, but is also higher cost.
The circuit diagram of a solar cell:
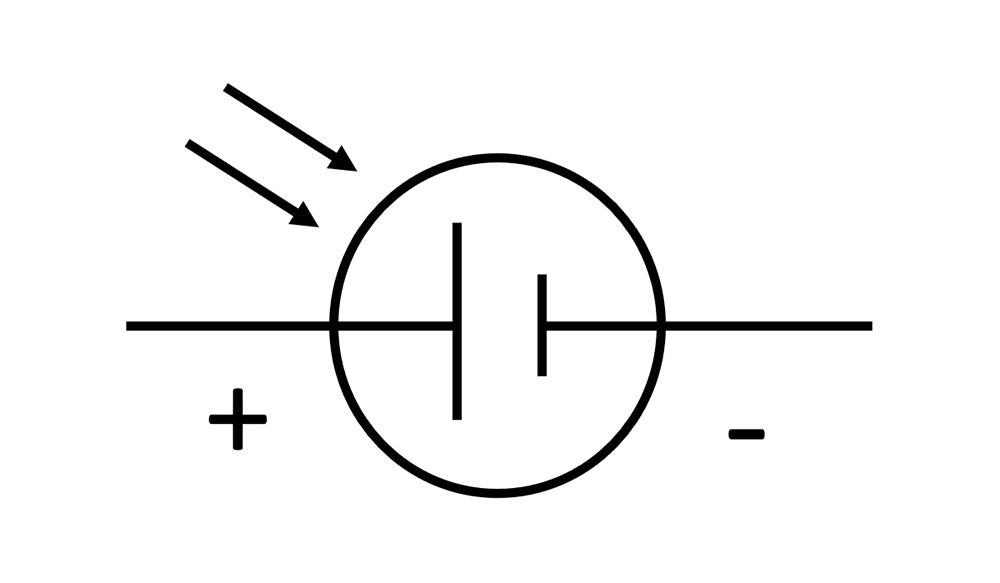
Combines elements of the circuit symbol for a battery, and the circuit symbol for a light sensitive device. When light hits the junction, the absorption of its energy liberates electrons, and these can flow to produce a current in a circuit. Diodes and transistors (both contain P-N junctions) are sensitive to light, hence most electronic components are packaged into light proof ‘chips’ as we want to keep the light out to prevent spurious operation.

The Raspberry Pi nicely illustrates this: The model 2 used a WL-CSP packaged chip (U16 in Figure 3), which has an exposed silicon die. This chip is designed for minimum size, and is usually used inside mobile phones, which have a light tight outer case. This chip was found to be sensitive to certain light sources, causing the Pi to crash. (See References)

A single silicon solar cell typically generates a maximum of about 0.6V, and only a small amount of current, which is not sufficient to power many devices. So solar cells are connected together (Figure 4) to create move useful voltages (such as 5V for a USB power pack).
The calculator in Figure 5 has 4 solar cells in its solar panel, which generate approximately 1.5V to power it.

Multiple solar cells are called a solar panel, and can come in many configurations of series and parallel connections to produce the required output voltage and current capacities. Often people refer to Panel and Cell interchangeably.
Using Solar Panels:
Modern solar cells have efficiencies ranging from around 10% to nearly 50%. A typical silicon cell has an average efficiency of about 20%. Solar cells only generate electricity when light is shining on them. It is common to store the energy generated so that it can be used when there is no light. When they are in the dark then no energy is generated, and due to leakage within the solar cell they can actually consume energy from an attached storage device (such as a battery or capacitor).
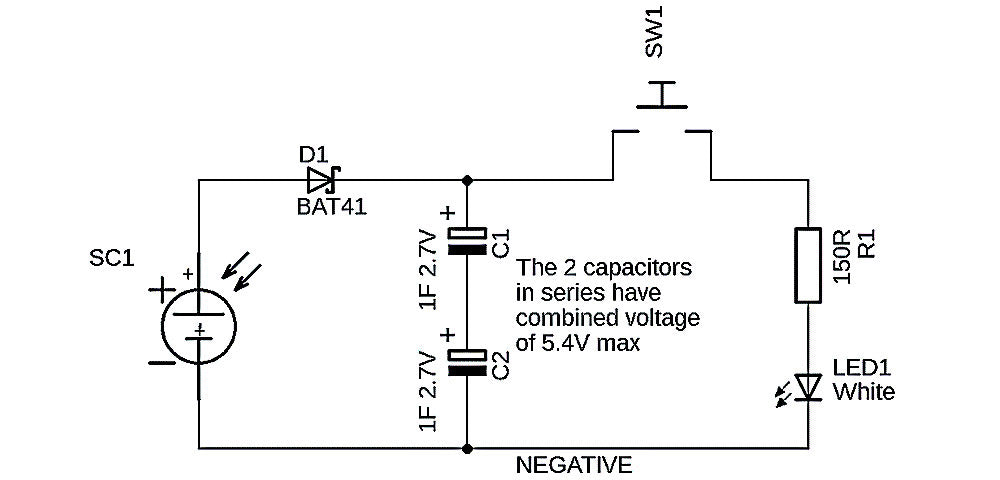
The Kitronik Solar Torch (2175) stores its energy in a pair of capacitors. Its circuit diagram is shown in Figure 6. The capacitors (C1 and C2) are charged by the solar cell, and then that energy can be used to light the LED. To prevent the solar cell ‘wasting’ the stored energy, there is a Schottky blocking diode in the circuit (D1). Schottky diodes have a low voltage drop, which means that less of the solar energy is wasted, and they are common in renewable power electronics.
Conclusions
Solar power has been around for a very long time, but it has only been of practical use since the invention of the semiconductor. Modern solar panels consist of a number of solar cells connected together to provide the required power output. Development in solar panels means that the efficiency is now up from around 1% to nearly 50% in the best cases, and the cost of generated electricity from solar cells is falling. Whilst solar cells only generate when exposed to light, it is possible to store that energy and use it later, for instance in a torch.
You can download a PDF of this guide here.
Images used in this document sourced from:
- Main Image: andreas160578 from Pixabay
- Figure 1: Charles frits first_solar_array-charles_fritts.jpg (800×608) (si-cdn.com) Viewed 11 Dec 2020
- Figure 2: Solar Cell circuit symbol by Kitronik
- Figure 3: Photo of Raspberry PI by Kitronik, Illustrates Raspberry PI Blog – https://www.raspberrypi.org/blog/xenon-death-flash-a-free-physics-lesson
- Figure 4: Photo of single solar cells. Wikipedia Solar cell – Solar cell – Wikipedia Viewed 16th Dec 2020
- Figure 5: Photo of solar powered calculator by Kitronik
- Figure 6: Solar Powered Torch schematic by Kitronik
If you enjoyed this guide, make sure you don’t miss out on any other new free learning resources by signing up for our newsletter here

